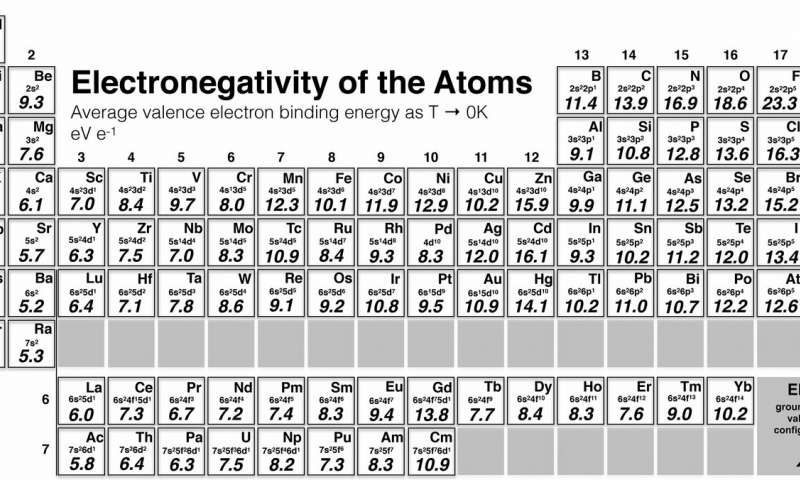
This is related to the hard and soft acids and bases principle 6. electronegativities are X A < X B < X D < X C). The additive quadratic form of ( 1) would allow powerful predictions to be made, for example, for exchange reactions: a reaction AB + CD = AC + BD would be energetically favorable when AC bond is the most polar, and BD bond is the least polar (i.e. Traditionally, any new scale, to be taken seriously, had to be consistent with Pauling’s. 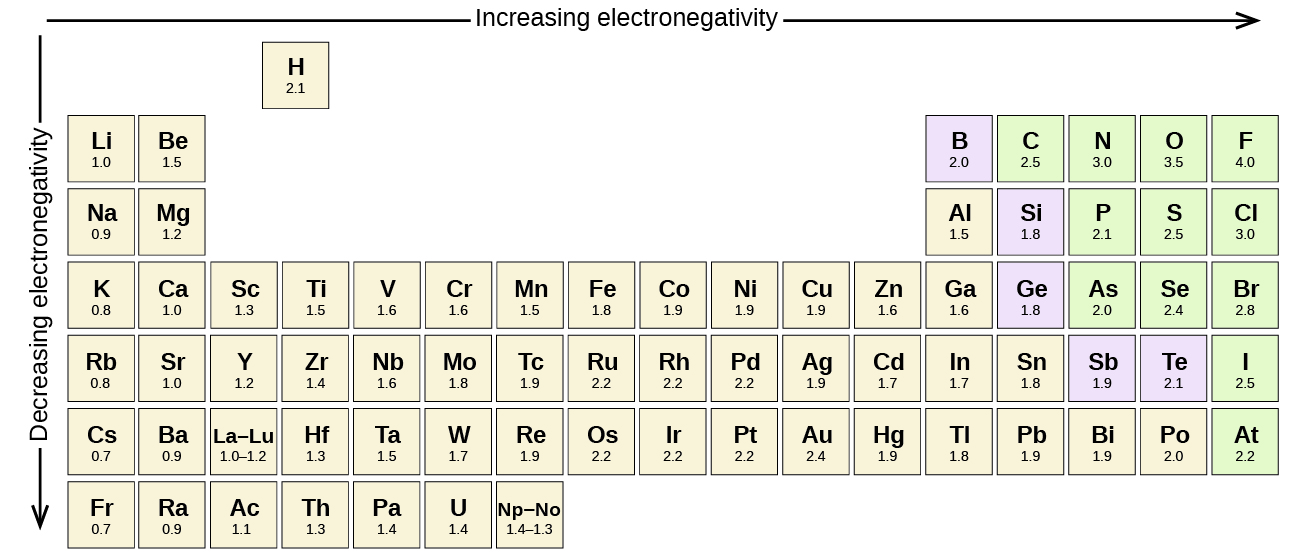
The resulting electronegativity scale has become the standard and enjoyed great success, remaining the most popular scale of electronegativity. This thermochemical scale was subsequently refined by Allred 5, who had more values of dissociation energies at his disposal, and more accurate values than at the time of original Pauling’s paper. Knowing that fluorine is the most electronegative element and arbitrarily setting its electronegativity to 4, Pauling obtained electronegativities of many elements using formula ( 1). Where D AB is the dissociation energy of a single chemical bond between two different atoms A and B, D AB cov is the covalent part of that dissociation energy modelled as D AA + D BB 2, and the amount of stabilization due to the ionic term equals just the electronegativity difference squared.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
